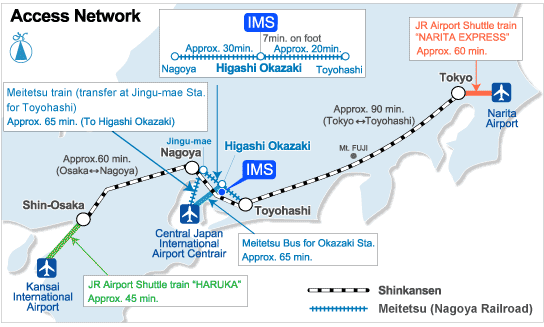
A novel organocatalytic strategy for the synthesis of highly substituted spirocyclopentaneoxindoles was developed employing simple nitrostyrenes and 3-substituted oxindoles as starting materials. Michael–Henry cascade reactions, enabled through cinchona alkaloid organocatalysis, provided products in high yield and excellent enantioselectivity in a single step.
Spirooxindoles in one step
If you just leaf through an organic chemistry journal, from JOC to Organic Letters, from EJOC to OBC, there is a huge number of papers describing synthesis of oxindoles! Indeed, the interest in the oxindoles scaffold rapidly increased in the last years and several methodologies have been published for the asymmetric synthesis of 3,3’-disubstituted oxindoles.
Cascade organacatalysis is a powerful tool for the for the asymmetric synthesis of very complex structures containing several stereogenic centers and oxindoles are optimal templates for the applications of these wonderful catalytic methodologies. For example, prof. Paolo Melchiorre, one the most expert in the organocatalysis field, published several works in the application of organocascade catalysis for the synthesis of quaternary oxindoles.
In this communication, Carlos Barbas, another pioneer of organocatalysis, reported a quindine-catalyzed synthesis of spirooxindoles through sequential Michael-Henry reactions.
They first performed a preliminary methodological study aimed at finding the most efficient catalyst, which turned out to be quindine derivative depicted below.
Subsequently the scope of the reaction has been expanded, by employing a wide range of substituted oxindoles, as well as different nitrostyrens. Performing the reaction at 0°C in CH2Cl2 as solvent in presence of quinidine derivative 1 permitted the isolation of the products in very high yield and optimal enantiomeric excess and diastereoisomeric ratio.
It is noteworthy to mention that this simple methodology permits the straightforward construction of the spirooxindole moiety containing four stereogenic centers,, with full control of the stereochemical outcome. This is another demonstration of the powerful of organocatalysis.