Multi-step synthesis using modular flow reactors: the preparation of yne-ones and their use in heterocycle synthesis
I.R. Baxendale, S.C. Schou, J. Sedelmeier, S.V. Ley, Chem. Eur. J. 2010, 16, 89-94.
http://onlinelibrary.wiley.com/doi/10.1002/chem.200902906/abstract

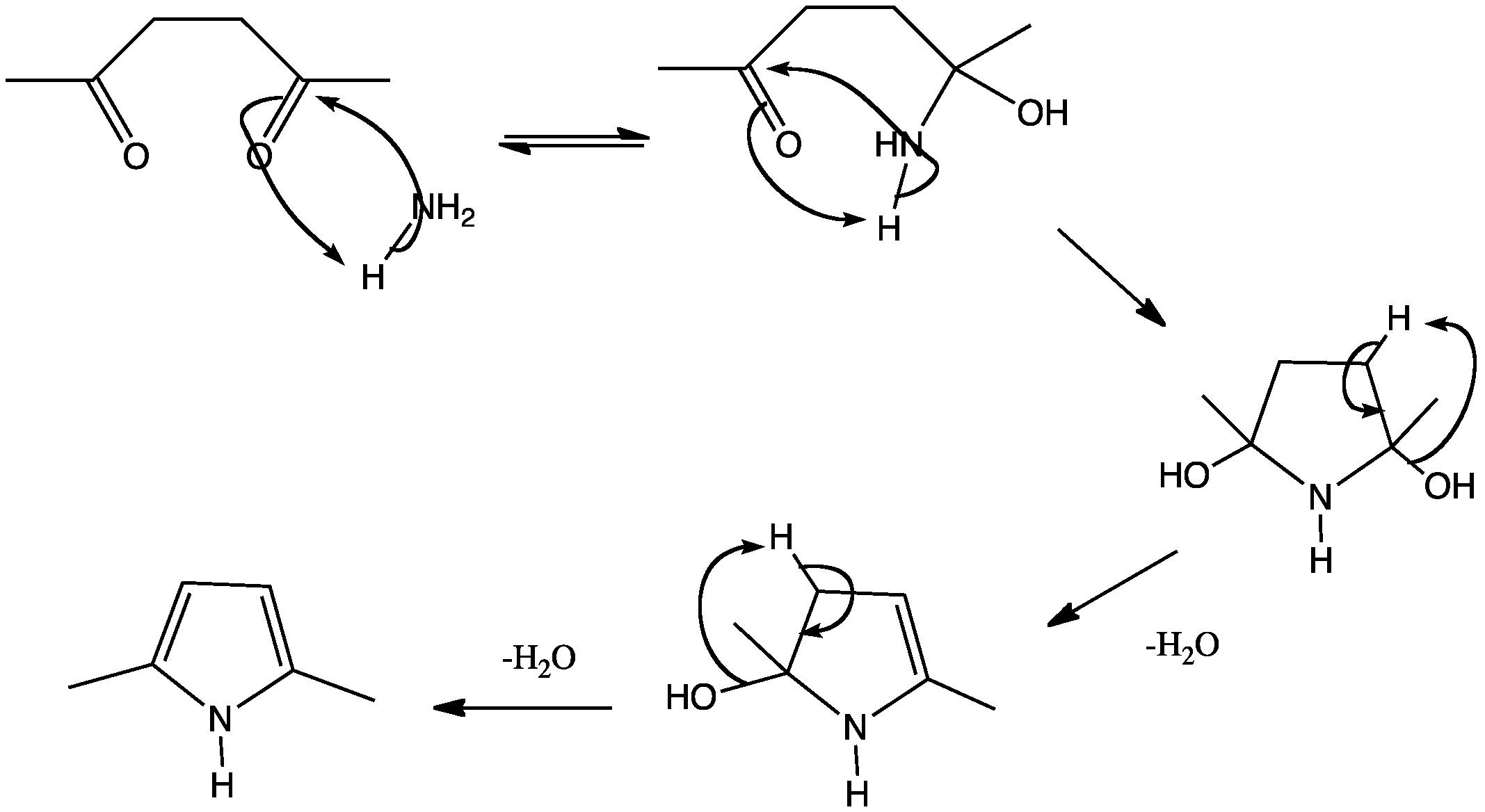
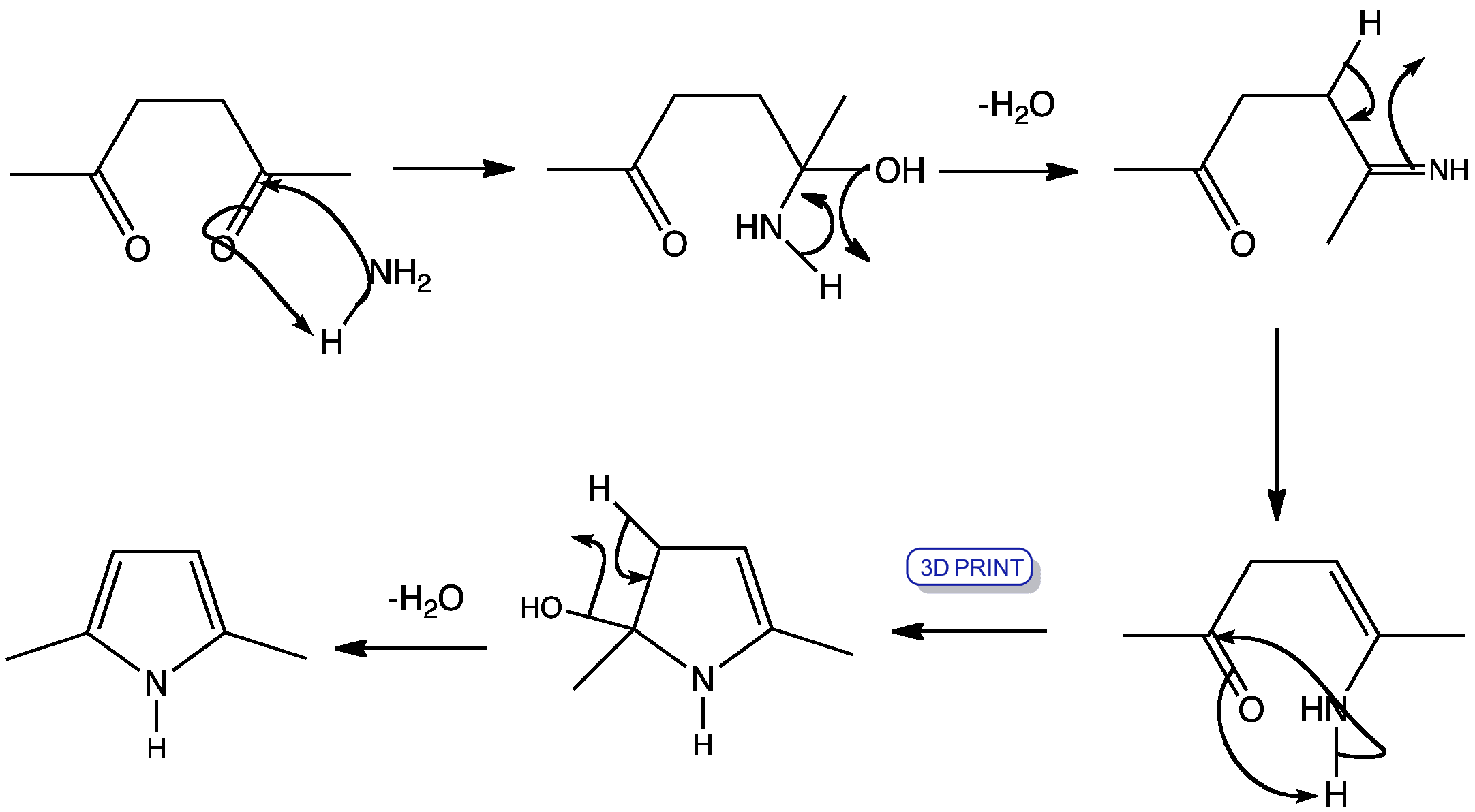
Multi-step in flow: The palladium-catalysed acylation of terminal alkynes for the synthesis of yne![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) ones as well as their further transformation to various heterocycles in a continuous-flow mode is presented. Furthermore, an extension of the simple flow configuration that allows for easy batch splitting and the generation of a heterocyclic library is described (see scheme).
ones as well as their further transformation to various heterocycles in a continuous-flow mode is presented. Furthermore, an extension of the simple flow configuration that allows for easy batch splitting and the generation of a heterocyclic library is described (see scheme).
![[BOND]](http://onlinelibrarystatic.wiley.com/undisplayable_characters/00f8ff.gif) ones as well as their further transformation to various heterocycles in a continuous-flow mode is presented. Furthermore, an extension of the simple flow configuration that allows for easy batch splitting and the generation of a heterocyclic library is described (see scheme).
ones as well as their further transformation to various heterocycles in a continuous-flow mode is presented. Furthermore, an extension of the simple flow configuration that allows for easy batch splitting and the generation of a heterocyclic library is described (see scheme).
 Methylene addends in fullerene electron acceptors increase
solar-cell efficiency
Methylene addends in fullerene electron acceptors increase
solar-cell efficiency
 Incorporation of a photosensitive azobenzene linker for
controllable carbohydrate orientation
Incorporation of a photosensitive azobenzene linker for
controllable carbohydrate orientation Halogen bonding drives the conversion from surface-confined
crystals to co-crystals without the use of solvent
Halogen bonding drives the conversion from surface-confined
crystals to co-crystals without the use of solvent