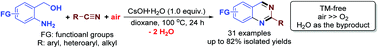http://pubs.rsc.org/en/Content/ArticleLanding/2017/GC/C7GC00977A?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+rss%2FGC+%28RSC+-+Green+Chem.+latest+articles%29#!divAbstract
Synthesis of 2-substituted quinazolines by CsOH-mediated direct aerobic oxidative cyclocondensation of 2-aminoarylmethanols with nitriles in air
Abstract
By using air as the superior oxidant, a highly atom-efficient synthesis of 2-substituted quinazolines is developed by a CsOH-mediated direct aerobic oxidative reaction of the readily available and stable 2-aminoarylmethanols and nitriles. Effectively working as the promoter in the alcohol oxidation, nitrile hydration, and cyclocondensation steps, CsOH is the best base for the reaction. A similar method can also be extended to the synthesis of substituted quinolines starting from methyl ketones instead of nitriles.
/////