EXAMPLE 2
(1) (2)
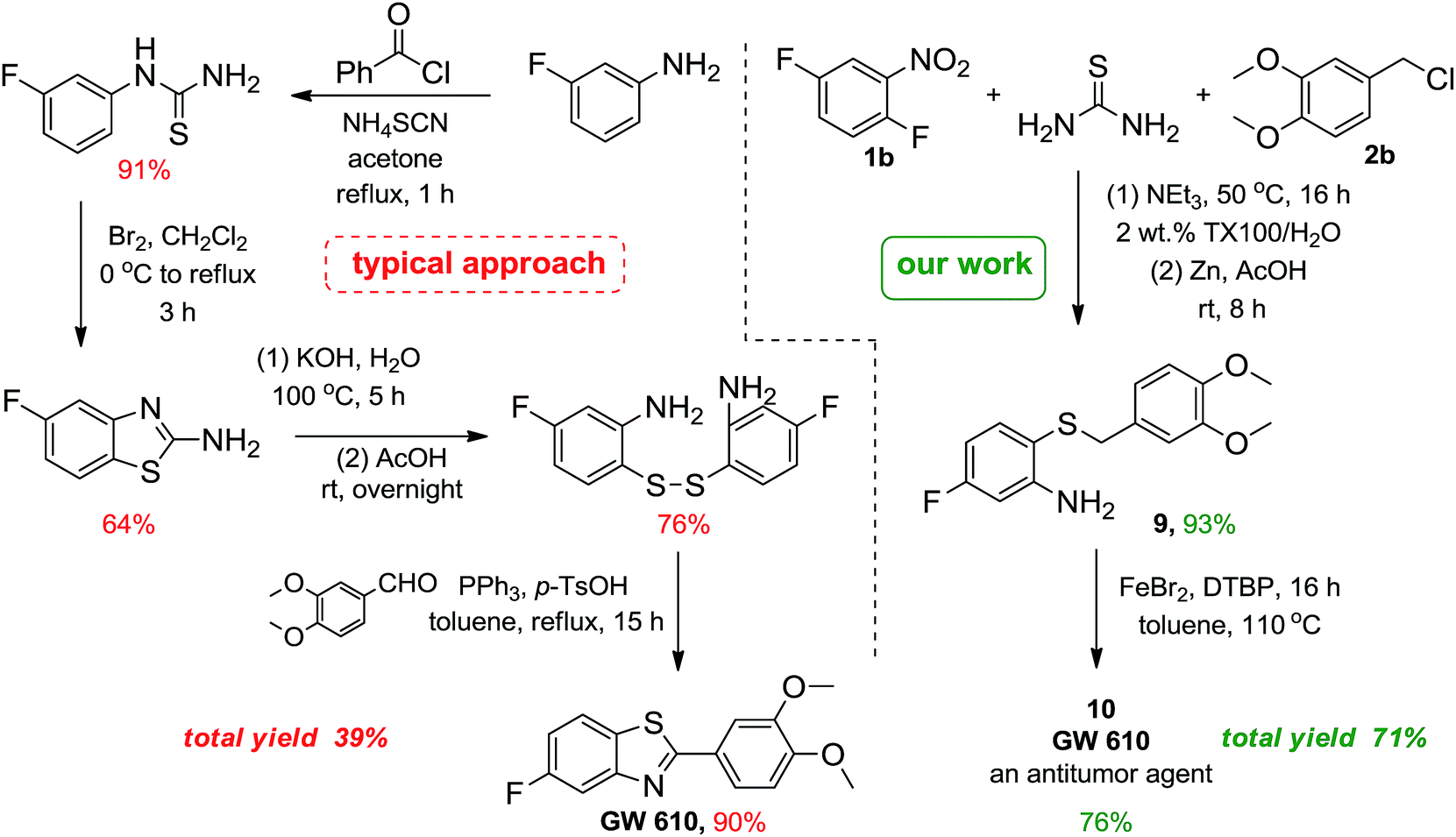
(iii), (iv)
(l-Aminomethyl-cyclobutyl)-acetic acid hydrochloride
Reagents: (i) Triethylphosphonoacetate, NaH; (ii) MeNO2, Bu4N+F"; (iii) H2, Ni; (iv) HCl
Synthesis of Cyclobutylidene-acetic acid ethyl ester (2)
NaH (60%) dispersion in oil, 1.80 g, 44.94 mmol) was suspended in dry tetrahydrofuran (80 mL) and cooled to 0°C. Triethylphosphonoacetate (9.33 mL, 47.08 mmol) was added and the mixture stirred at 0°C for 15 minutes. Cyclobutanone (1) (3.0 g, 42.8 mmol) in THF (20 mL) was then added and the mixture allowed to warm to room temperature. After 2 hours, the mixture was partitioned between diethyl ether (200 mL) and water (150 mL). The organic phase was separated, washed with brine, dried (MgSO4), and the solvent removed in vacuo at 600 mm Hg. The residue was purified by flash chromatography (silica, ethyl acetate :pentane 1 : 19) to give 5.81 g (96%) of (2) as a colorless oil. iH NMR, 400 MHz (CDCI3): δ 1.27 (3H, t, J=6Hz), 2.09 (2H, m), 2.82 (2H, m),
3.15 (2H, m), 4.14 (2H, q, J = 6 Hz), 5.58 (IH, s).
MS (ES+) m/e: 141 ([MH+], 100%). IR (film) v cm"1: 1088, 1189, 1336, 1673, 1716,2926. Synthesis of (l-Nitromethyl-cyclobutyl)-acetic acid ethyl ester (3)
The unsaturated ester (2) (5.79 g, 41.4 mmol) was dissolved in tetrahydrofuran (20 mL) and stirred at 70°C with nitromethane (4.67 mL, 86.4 mmol) and tetrabutylammonium fluoride (1.0 M in tetrahydrofuran, 55 mL, 55.0 mmol). After 18 hours, the mixture was cooled to room temperature, diluted with ethyl acetate (150 mL), and washed with 2N HCl (60 mL) followed by brine (100 mL). The organic phase was collected, dried (MgSO4), and the solvent removed in vacuo. The residue was purified by flash chromatography (silica, ethyl acetate:heptane 1 :1) to give 4.34 g (52%) of a clear oil. !H NMR 400 MHz (CDC13): δ 1.27 (3H, t, J = 6 Hz), 1.96-2.20 (6H, m), 2.71
(2H, s), 4.15 (2H, q, J = 6 Hz), 4.71 (2H, s).
MS (ES+) m/e: 202 ([MH+], 100%).
IR Cfiln- v cm-1 : 1189, 1378, 1549, 1732, 2984.
Synthesis of (l-Aminomethyl-cyclobutyl)-acetic acid hydrochloride (4) The nitroester (3) (2.095 g, 10.4 mmol) was dissolved in methanol
(50 mL) and shaken over Raney nickel catalyst under an atmosphere of hydrogen (45 psi) at 30°C. After 6 hours, the catalyst was removed by filtration through celite. The solvent was removed in vacuo to give 1.53 g of a pale yellow oil which was used without purification. The oil was dissolved in 1 ,4-dioxane (5 mL) and 6N HCl (15 mL) and heated to reflux. After 5 hours, the mixture was cooled to room temperature, diluted with water (20 mL), and washed with dichloromethane (3 x 30 mL). The aqueous phase was collected and the solvent removed in vacuo. The residue was triturated with ethyl acetate to give 1.35 g (72%) of a white solid after collection and drying. !H NMR 400 MHz (de-DMSO): δ 1.80-2.03 (6H, m), 2.59 (2H, s), 3.02 (2H, s),
8.04 (3H, br s), 12.28 (IH, br s).
MS (ES+) m/e: 144 ([MH-HC1J+, 100%). Microanalysis calculated for C7H14NO2CI:
C, 46.80%; H, 7.86%; N, 7.80%. Found: C, 46.45%; H, 7.98%; N, 7.71%.
//////////////http://www.google.co.in/patents/WO1999021824A1?cl=en





 .
.