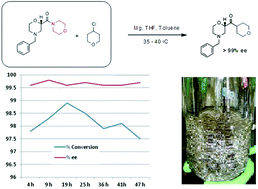
A key pharmaceutical intermediate (
1) for production of edivoxetine·HCl was prepared in >99% ee
via
a continuous Barbier reaction, which improves the greenness of the
process relative to a traditional Grignard batch process. The Barbier
flow process was run optimally by Eli Lilly and Company in a series of
continuous stirred tank reactors (CSTR) where residence times,
solventcomposition, stoichiometry, and operations temperature were
optimized to produce 12 g h
−1crude ketone
6 with 98% ee and 88%
in situ yield for 47 hours total flow time. Continuous salt formation and isolation of intermediate
1 from the ketone solution was demonstrated at 89% yield, >99% purity, and 22 g h
−1
production rates using MSMPRs in series for 18 hours total flow time.
Key benefits to this continuous approach include greater than 30%
reduced process mass intensity and magnesium usage relative to a
traditional batch process. In addition, the flow process imparts
significant process safety benefits for Barbier/Grignard processes
including >100× less excess magnesium to quench, >100× less
diisobutylaluminum hydride to initiate, and in this system, maximum
long-term scale is expected to be 50 L which replaces 4000–6000 L batch
reactors.
A continuous flow Barbier reaction was employed for the production of a key pharmaceutical intermediate (
1) in the synthesis of edivoxetine·HCl (a highly selective norepinephrine re-uptake inhibitor).

US scientists from Eli Lilly and Company and D&M Continuous Solutions, led by
Michael Kopach,
report the development of a continuous Barbier reaction which preserves
chirality and the product obtained in >99% ee. The team ran the
process in a series of continuous stirred tank reactors, where residence
time, solvent composition, stoichiometry and operations temperature
were optimised to produce 12 g per hour of the ketone precursor to
1 with 98% ee and 88%
in situ yield for 47 hours total flow time. Continuous salt formation and isolation of
1 could then be achieved from the ketone solution with >99% purity.
This
process offers up several significant advantages over a traditional
Grignard batch process. This continuous flow method gave greater than
30% reduced process mass intensity and magnesium usage relative to the
batch method. Equally, the flow process resulted in >100 x less
excess magnesium to quench and >100 x less diisobutylaluminum hydride
to initiate giving significant safety benefits. The authors expect
that the maximum long-term scale of the process is 50 L which would
replace 4000-6000 L batch reactors.
Continuous Flow Barbier Reaction
Figure 2. Continuous Barbier Laboratory Setup
For
100 years, Grignard reactions have been one of the most powerful and
effi cient organic chemistry methodologies for C-C bond formation.
However, Grignard reactions are also among the most challenging
reactions from both operational and potential safety issues due to
initiation diffi culties and runaway potential. A close variation to the
Grignard reaction is the Barbier reaction wherein the Grignard reagent
is prepared in the presence of an electrophile resulting in the
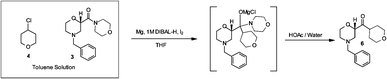
immediate consumption of the Grignard. A Barbier reaction using a CSTR
was developed for a key pharmaceutical intermediate in production of
edivoxetine·HCl (
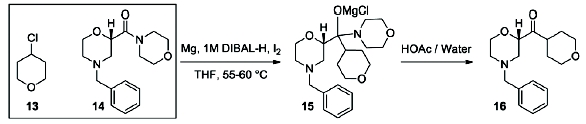
Scheme 4) [9]. In the fl ow setup (
Figure 2),
solid magnesium is sequestered in the fi rst tank where the Grignard
initiation event takes place. CSTR 2 was used as an aging tank and CSTR 3
was the quench tank. CSTRs were used for Grignard reaction rather than a
PFDR because of the solid Mg reagent.
Scheme 4: Barbier Reaction to form Ketone 15
Continuous
reaction improved process safety, product quality, and process
greenness. The continuous reaction achieved >99% ee in situ versus
95% ee batch because of immediate conversion of unstable intermediate.
Solvent volumes were reduced 30%. The safety hazards were reduced by
decreasing the reactor size by 50X, which reduced chemical potential and
also increased heat transfer surface area per unit volume by 4X.
DIBAL-H initiating agent was reduced by more than 100X, and excess Mg
that must be quenched at the end of reaction was almost eliminated. When
run continuously, the commercial scale Grignard formation reactor was
expected to be 50L, which replaces 4000-6000L batch reactor.
The continuous flow Barbier reaction: an improved environmental alternative to the Grignard reaction?
bD&M Continuous Solutions, Indianapolis, USA
Green Chem., 2012,14, 1524-1536
DOI: 10.1039/C2GC35050E
http://pubs.rsc.org/en/Content/ArticleLanding/2012/GC/C2GC35050E#!divAbstract
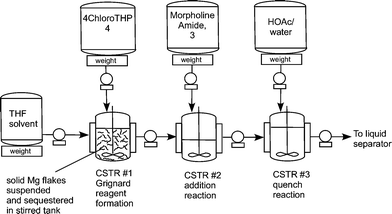
Three vessel Grignard CSTR process train.
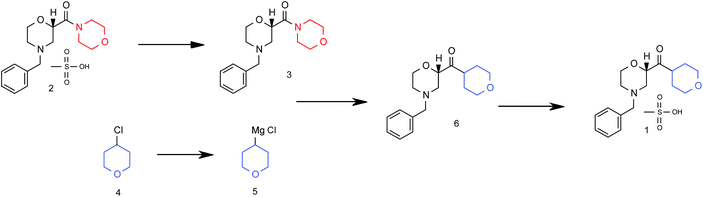
Grignard synthesis of compound 1.
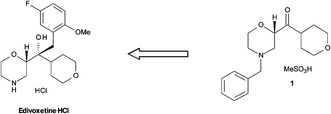
Retrosynthesis of edivoxetine·HCl.
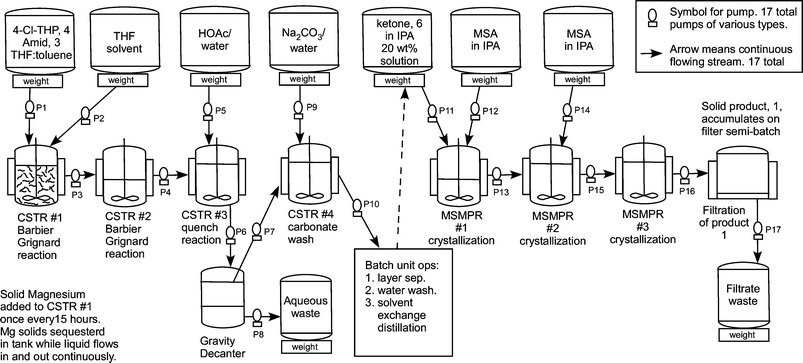
Flow diagram for the whole continuous process from amide 3 to product 1.
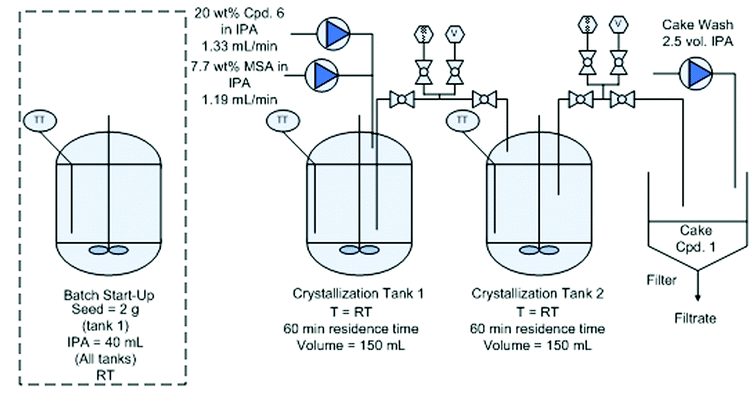
Continuous crystallization of compound 1.

Distillation and continuous crystallization of compound 1.
Entry, Rxn temp. (°C), Vol. ratio THF–toluene (%), Conversion (%), ee (%)
//////////The continuous flow, Barbier reaction, improved environmental alternative, Grignard reaction, FLOW SYNTHESIS
























 Flow diagram for the whole continuous process from amide 3 to product 1.
Flow diagram for the whole continuous process from amide 3 to product 1.





