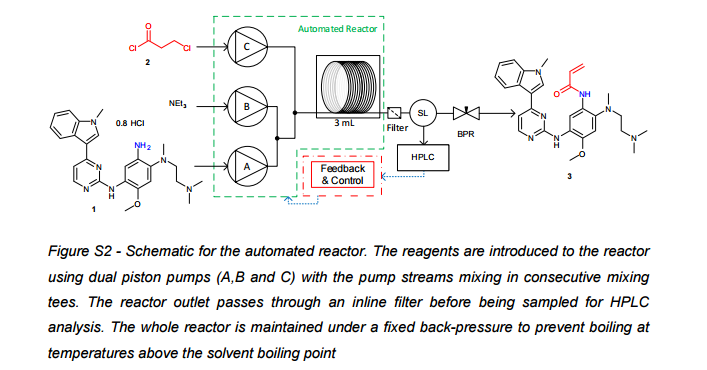
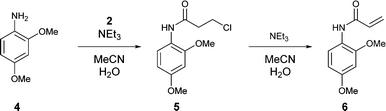
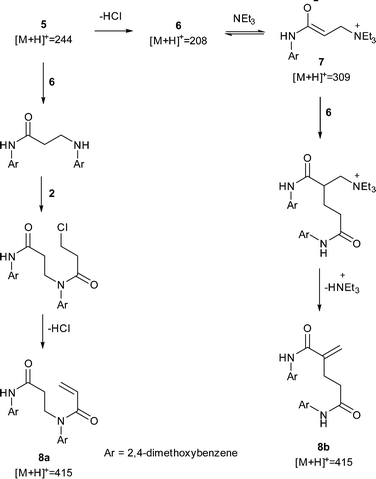
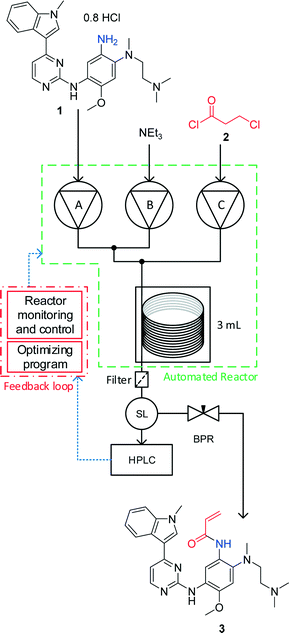
.
.In search of developing new useful “click reactions”, herein we report the organocatalytic azomethine imine-olefin [3 + 2]-cycloaddition as a new click reaction for the synthesis of drug-like spiroindane-1,3-dione-pyrazolidinones from indane-1,3-diones, aldehydes and N,N-cyclic azomethine imines through amino acid-catalysis. The scope of this new click reaction is demonstrated using many examples with high reactivity, selectivity and yields.



Happy to share with you our recent work on the development of Tomita zipper-cyclization (TZC) reaction…I am sure that this reaction may become useful tool for organic chemists in near future…
- Catalysis Laboratory, School of Chemistry, University of Hyderabad
- Hyderabad, India 500 046
Paper
Organocatalytic azomethine imine-olefin click reaction: high-yielding stereoselective synthesis of spiroindane-1,3-dione-pyrazolidinones
*Corresponding authors
a
School of Chemistry, University of Hyderabad, Hyderabad-500 046, India
E-mail: ramsc@uohyd.ac.in
Fax: +91-40-23012460
E-mail: ramsc@uohyd.ac.in
Fax: +91-40-23012460
Org. Biomol. Chem., 2016, Advance Article
DOI: 10.1039/C6OB01009A http://pubs.rsc.org/en/content/articlelanding/2016/ob/c6ob01009a#!divAbstract
//////////















 .
.


 .
.









 Open Access
Open Access