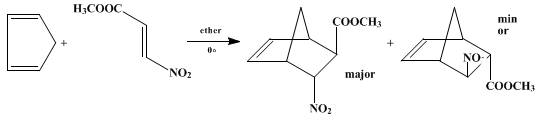
Methyl (E)-3-nitroacrylate is a reactive dienophile in the Diels-Alder reaction. With cyclopentadiene it gives the corresponding adduct, methyl 6-nitro-2-norbornene-5-carboxylate, in 43% yield in refluxing benzene9 or in quantitive yield in ether at 0°,10 as shown below. The adduct was shown by NMR analysis to consist of an 86 : 14 mixture of endo- : exo-nitro stereoisomers, which could not be separated by crystallization.10
METHYL (E)-3-NITROACRYLATE
[2-Propenoic acid, 3-nitro-, methyl ester (E)-]
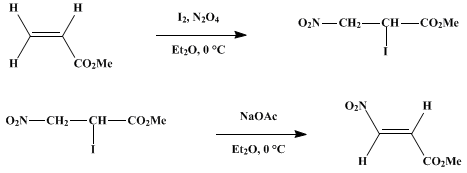 |
1H NMR (60 MHz, CDCl3): δ 3.8 (s, 3H, OCH3), 6.95 (d, J = 14 hz, 1H, =CHNO2-), 7.55 (d, J = 14 Hz, 1H, =CHNO2): IR (CCl4): cm.−1 3100 s, 3000 m, 2950 s, 2880 m (all CH), 1720 s (C=O), 1640 ms (C=C), 1530 s and 1350 s (NO2).