Green Chem., 2016, Advance Article
DOI: 10.1039/C5GC03102H, Paper
L. Puchot, P. Verge, T. Fouquet, C. Vancaeyzeele, F. Vidal, Y. Habibi
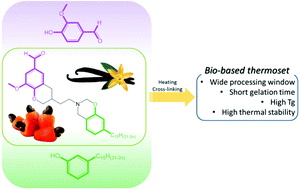
Asymmetric di-benzoxazine monomers from naturally occurring phenolic compounds - cardanol and vanillin - were synthesized to obtain a processable and self-supported bio-thermoset with valuable properties. Such strategy constitutes an efficient and versatile route for the elaboration of biobased thermoset from a wide range of phenolic compounds derived from renewable resources.
http://pubs.rsc.org/en/Content/ArticleLanding/2016/GC/C5GC03102H?utm_source=feedburner&utm_medium=feed&utm_campaign=Feed%3A+rss%2FGC+%28RSC+-+Green+Chem.+latest+articles%29#!divAbstract
With the ongoing efforts to promote the development of bio-based dibenzoxazine thermosets, we explore herein a new strategy aiming at the synthesis of asymmetric dibenzoxazine monomers from naturally occurring phenolic compounds, cardanol and vanillin. By taking advantage of the low reactivity of cardanol, a monosubstituted cardanol-based benzoxazine monomer was prepared and further coupled with vanillin to yield vanillin–cardanol di-benzoxazines. The structural features of the resulting products were substantiated by 1H NMR and HR-MS. The occurrence of the thermally-induced ring-opening polymerization was monitored by rheological measurements and DSC. At 190 °C the new asymmetric monomers showed a moderate gelation time (8 min) compared to 30–31 min revealed for cardanol-based (di-card) dibenzoxazines. Once polymerized, they exhibited a high Tg (129 °C), while the di-card flew under heating because of its low cross-linking density. Asymmetric monomers also exhibited lower melting temperatures than their symmetrical congeners based on vanillin, which significantly enlarge the processing window between the melting and polymerization temperatures up to 126 °C instead of 7 °C for symmetric vanillin-based dibenzoxazines. Therefore, such a strategy constitutes an efficient and versatile route for an easy elaboration of biobased monocomponent thermosets and can be applied to a wide range of phenolic compounds derived from renewable resources.
Breaking the symmetry of dibenzoxazines: a paradigm to tailor the design of bio-based thermosets
b
Laboratoire de Physicochimie des Polymères et des Interfaces (LPPI – EA 2528), I-Mat, Université de Cergy-Pontoise, 5 mail Gay-Lussac, 95031 Cergy-Pontoise, France
c
Environmental Measurement Technology Group, Environmental Management and Research Institute (EMRI), National Institute of Advanced Industrial Science and Technology (AIST), Onogawa 16-1, Tsukuba, Japan
Green Chem., 2016, Advance Article
DOI: 10.1039/C5GC03102H //////////
////////