React. Chem. Eng., 2016, Advance Article
DOI: 10.1039/C6RE00059B, Paper

Open Access
Nicholas Holmes, Geoffrey R. Akien, A. John Blacker, Robert L. Woodward, Rebecca E. Meadows, Richard A. Bourne
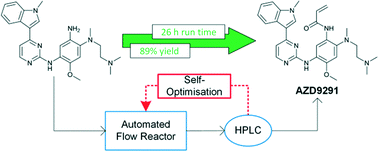
Self-optimising flow reactors combine online analysis with evolutionary
feedback algorithms to rapidly achieve optimum conditions.
Self-optimising
flow reactors combine online analysis with evolutionary feedback
algorithms to rapidly achieve optimum conditions. This technique has
been applied to the final bond-forming step in the synthesis of AZD9291,
an irreversible epidermal growth factor receptor kinase inhibitor
developed by AstraZeneca. A four parameter optimisation of a telescoped
amide coupling followed by an elimination reaction was achieved using
at-line high performance liquid chromatography. Optimisations were
initially carried out on a model compound (2,4-dimethoxyaniline) and the
data used to track the formation of various impurities and ultimately
propose a mechanism for their formation. Our protocol could then be
applied to the optimisation of the 2-step telescoped reaction to
synthesise AZD9291 in 89% yield.
Paper
Self-optimisation of the final stage in the synthesis of EGFR kinase inhibitor AZD9291 using an automated flow reactor
aInstitute of Process Research and Development, School of Chemistry, University of Leeds, Leeds, UK
E-mail: r.a.bourne@leeds.ac.uk
bDepartment of Chemistry, Faraday Building, Lancaster University, Lancaster, UK
cSchool of Chemical and Process Engineering, University of Leeds, Leeds, UK
dAstraZeneca Pharmaceutical Development, Silk Road Business Park, Macclesfield, UK
React. Chem. Eng., 2016, Advance Article
DOI: 10.1039/C6RE00059B
http://pubs.rsc.org/en/Content/ArticleLanding/2016/RE/C6RE00059B#!divAbstract

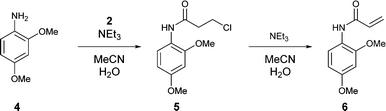
| | Scheme 1 Synthesis of the model acrylamide 6 via the β-chloroamide 5 intermediate. |

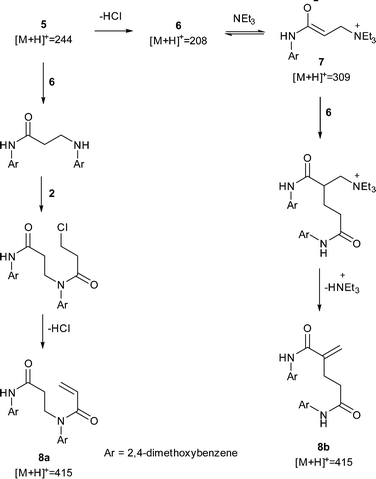
| | Scheme 2 Proposed mechanisms to dimers 8a and 8b. The observation of a peak corresponding to 7suggested a Rauhut–Currier mechanism to 8b but subsequent LC-MS-MS analysis showed the major dimer to most likely be 8a. All observed peaks from offline LC-MS are displayed. |
 ////
///////Self-optimisation, synthesis, EGFR kinase inhibitor, AZD9291, automated flow reactor




 Open Access
Open Access