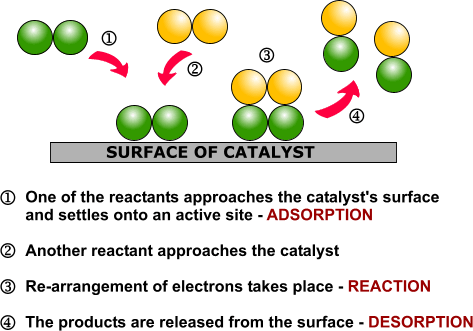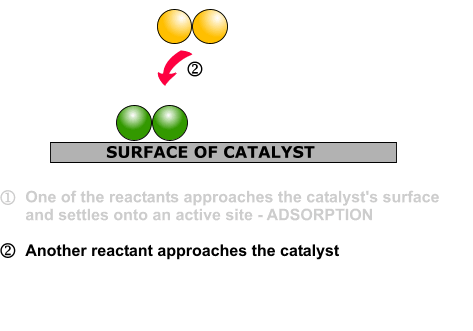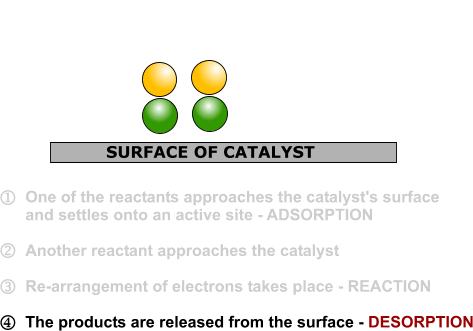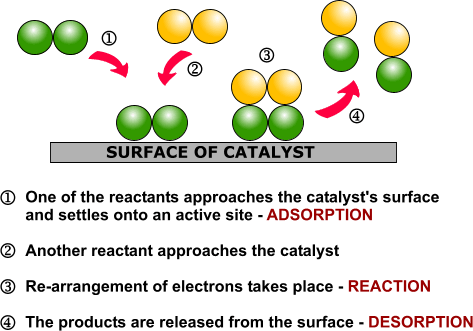
Heterogeneous catalysis and catalyst recycling
Heterogeneous
catalysis is a type of catalysis in which the catalyst occupies a
different phase from the reactants and products. This may refer to the
physical phase — solid, liquid or gas — but also to immiscible fluids.
Heterogeneous catalysts can be more easily recycled than homogeneous,
but characterization of the catalyst and optimization of properties can
be more difficult.



Heterogeneous
catalysis is widely used in the synthesis of bulk and fine chemicals.
In a general, small scale batch reaction, the catalyst, reactants,
and solvent are stirred together until completion of the reaction, after
which the bulk liquid is separated by filtration. The catalyst can then
be collected for either recycling or disposal. In a continuous process,
the catalyst can be fixed in space and the reaction mixture allowed to
flow over it. The reaction and separation are thus combined in a single
step, and the catalyst remains in the reactor for easy recycling. Beyond
facilitating separation, thecatalyst may have improved lifetime due to
decreased exposure to the environment, and reaction rates and turnover
numbers can be enhanced through the use of high concentrations of
a catalyst with continuous recycling. The benefits of flow are seemingly
obvious, yet it has only recently become a widely adopted method for
bench-scale synthesis.
1


The most common application of continuous heterogeneous catalysis is in hydrogenation reactions,
2 where
the handling and separation of solid precious metal catalysts is not
only tedious but hazardous under batch conditions. Moreover, the mixing
between the three phases in a hydrogenation is generally quite poor. The
use of a flow reactor gives a higher interfacial area between phases
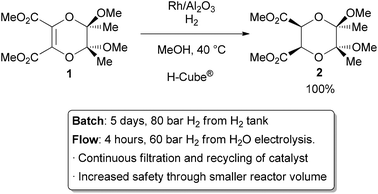
and thus more efficient reactions. For example, Ley and co-workers found
that the hydrogenation of alkene
1 to
2 was challenging in batch, requiring multiple days at 80 bar of H
2 (Scheme 1).
3 Using
a commercially available H-Cube® reactor, the reaction time was
shortened to 4 hours, the pressure reduced to 60 bar, and manual
separation and recycling of the catalyst from the reaction was
unnecessary. The increased efficiency is due to a combination of
improved mixing of the three phases, as well as the continuous recycling
and high local concentration of the catalyst. The H-Cube offers a
further safety advantage because it generates hydrogen gas on demand
from water, obviating the need for a high pressure H
2 tank.

 |
| | Scheme 1 Hydrogenation with an immobilized heterogeneous catalyst. | |
Homogeneous
catalysis has many advantages over heterogeneous catalysis, such as
increased activity and selectivity, and mechanisms of action that are
more easily understood. Unfortunately, the difficulty associated with
separating homogeneous catalysts from the product is a significant
hindrance to their large scale application. In an attempt to combine the
high activity of homogeneous catalysis with the practical advantageous
of heterogeneous catalysis, there has been much research into
immobilizing homogeneous catalysts on solid supports.
4 This
is generally achieved by linking thecatalyst to the surface of an
insoluble solid such as silica or polymer beads. As was the case in
batch hydrogenation reactions, the process of separating and purifying
the catalyst is inefficient, potentially dangerous, and may lead to
degradation and loss of material. Performing these reactions in a flow
system can help overcome these problems.
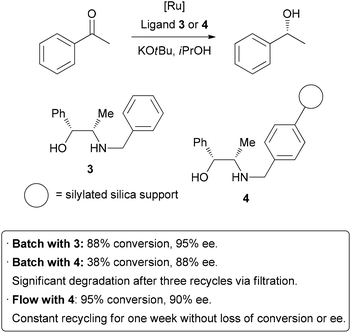
5 A highly efficient
example has been demonstrated by van Leeuwen and co-workers, who sought
to immobilize a catalyst used in transfer hydrogenation reactions
(Scheme 2).
6Their test reaction was the asymmetric reduction of acetophenone; homogeneousreduction with ruthenium and ligand
3 provided
88% conversion and 95% enantioselectivity. The ligand was then
covalently linked to silica gel through the benzyl group to form
4.
Using this heterogenized system under batch conditions, conversion
dropped to 38% on the same time scale, and a slight decrease in
enantioselectivity occurred. A reduction in activity of a catalyst upon
immobilization is common, so highly efficient recycling is required.
Unfortunately, when attempting to re-use the catalyst after filtration,
significant degradation and leaching occurred. The catalyst was then
packed in a glass column for application in flow chemistry. After a
short optimization of flow rate, 95% conversion and 90% ee were
obtained. Importantly, the reaction could be run continuously for up to
one week without significant degradation in conversion or
enantioselectivity. The physical isolation of catalyst species on the
solid support is suggested to contribute to the long catalystlifetime.
Interestingly, the basic potassium tert-butoxide additive was only
required initially to activate the catalyst, and the reaction could
subsequently be run without additional base, allowing the product to be
isolated completely free of additives. It is important to note, on top
of the decreased activity due to modification, that leaching from
cleavage off the solid support and the increased cost of
the catalyst due to derivatization are all potential downsides of
immobilization of catalysts. In some instances, a seemingly
heterogeneous catalyst has been shown to leach active homogeneous
species into solution.
7 However, as can be seen above, robust
systems can be developed which do combine the best features of both
homogeneous and heterogeneous catalysis.

 |
| | Scheme 7 Immobilization of a homogeneous catalyst on a solid support. | |
Another
important method for recycling expensive catalysts is through the use
of liquid–liquid biphasic conditions where the catalyst and reactants
can be separated by extraction upon completion of the reaction. Such
processes have already been utilized on the medium and large scale in a
continuous or semi-continuous fashion.
8,9 Recycling on a
small scale is typically done through batch liquid–liquid extractions,
but examples using continuous methods are increasing.
10-13 A
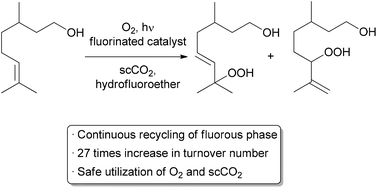
recent automated small scale recycling of a biphasic catalyst system was
demonstrated by the George group in the continuous oxidation
of citronellol (Scheme 3).
14A highly fluorinated porphyrin was used as the photocatalyst, and a combination of hydrofluoroether (HFE) and scCO
2 was
used as the solvent. Under high pressure flow conditions, a single
phase was observed. Depressurization occurred after the reactor,
resulting in two phases – the organic product in one, and
the catalyst and HFE in the other. The denser, catalyst-containing
fluorous phase was continuously pumped back through the reactor. With
this method, the catalyst was recycled 10 times while maintaining 75% of
its catalytic activity, giving an increase in TON of approximately
27-fold compared to previous batch conditions. Some leaching of the
fluorinated catalyst into the organic product was observed, accounting
for the decreased activity over time.

 |
| | Scheme 3 Automated recycling of a biphasic catalyst system. |

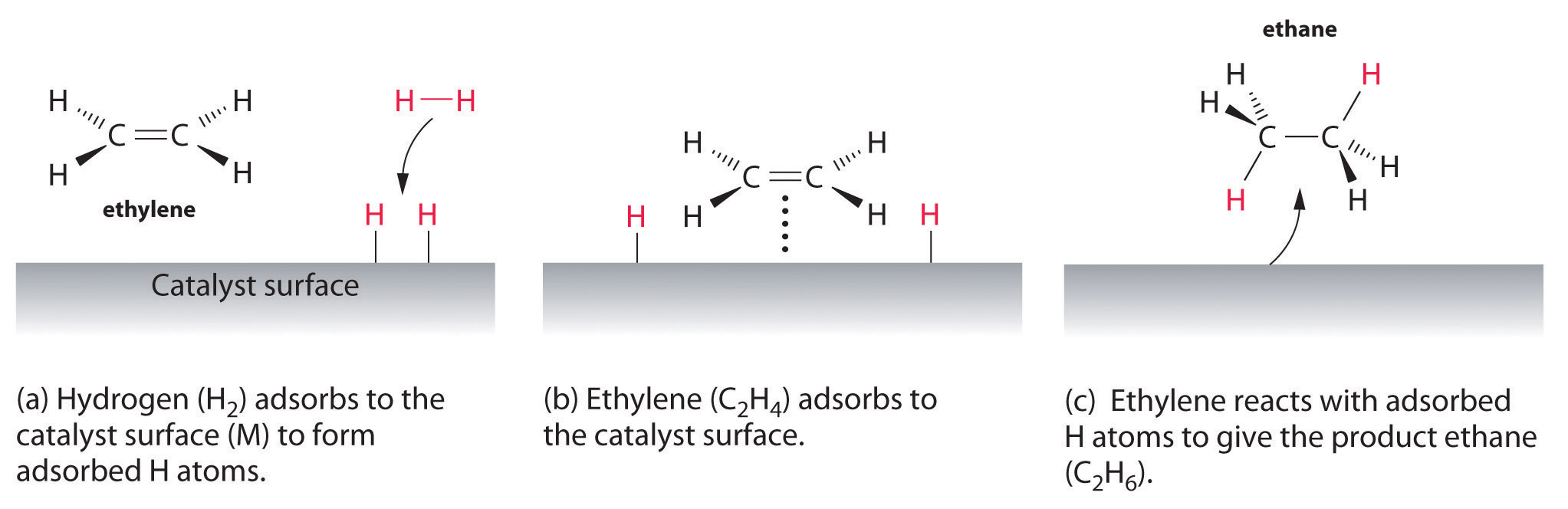
Examples of heterogeneous catalysisThe hydrogenation of a carbon-carbon double bondThe simplest example of this is the reaction between ethene and hydrogen in the presence of a nickel catalyst.  In
practice, this is a pointless reaction, because you are converting the
extremely useful ethene into the relatively useless ethane. However, the
same reaction will happen with any compound containing a carbon-carbon
double bond.One important industrial use is in the hydrogenation of
vegetable oils to make margarine, which also involves reacting a
carbon-carbon double bond in the vegetable oil with hydrogen in the
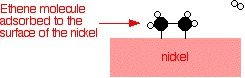
presence of a nickel catalyst.Ethene molecules are adsorbed on the
surface of the nickel. The double bond between the carbon atoms breaks
and the electrons are used to bond it to the nickel surface.
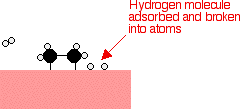
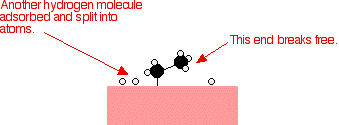
Hydrogen
molecules are also adsorbed on to the surface of the nickel. When this
happens, the hydrogen molecules are broken into atoms. These can move
around on the surface of the nickel. In
practice, this is a pointless reaction, because you are converting the
extremely useful ethene into the relatively useless ethane. However, the
same reaction will happen with any compound containing a carbon-carbon
double bond.One important industrial use is in the hydrogenation of
vegetable oils to make margarine, which also involves reacting a
carbon-carbon double bond in the vegetable oil with hydrogen in the
presence of a nickel catalyst.Ethene molecules are adsorbed on the
surface of the nickel. The double bond between the carbon atoms breaks
and the electrons are used to bond it to the nickel surface.
Hydrogen
molecules are also adsorbed on to the surface of the nickel. When this
happens, the hydrogen molecules are broken into atoms. These can move
around on the surface of the nickel.
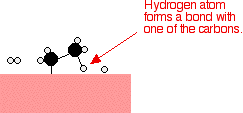
If
a hydrogen atom diffuses close to one of the bonded carbons, the bond
between the carbon and the nickel is replaced by one between the carbon
and hydrogen.
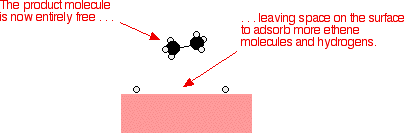
That end of the original ethene now breaks free of the surface, and eventually the same thing will happen at the other end.
As
before, one of the hydrogen atoms forms a bond with the carbon, and
that end also breaks free. There is now space on the surface of the
nickel for new reactant molecules to go through the whole process again.
Catalytic converters
Catalytic
converters change poisonous molecules like carbon monoxide and various
nitrogen oxides in car exhausts into more harmless molecules like carbon
dioxide and nitrogen. They use expensive metals like platinum,
palladium and rhodium as the heterogeneous catalyst.
The metals
are deposited as thin layers onto a ceramic honeycomb. This maximises
the surface area and keeps the amount of metal used to a minimum.
Taking the reaction between carbon monoxide and nitrogen monoxide as typical:
  |
| | |
Catalytic converters can be affected by catalyst poisoning.
This happens when something which isn't a part of the reaction gets
very strongly adsorbed onto the surface of the catalyst, preventing the
normal reactants from reaching it.Lead is a familiar catalyst poison for
catalytic converters. It coats the honeycomb of expensive metals and
stops it working.In the past, lead compounds were added to petrol
(gasoline) to make it burn more smoothly in the engine. But you can't
use a catalytic converter if you are using leaded fuel. So catalytic
converters have not only helped remove poisonous gases like carbon
monoxide and nitrogen oxides, but have also forced the removal of
poisonous lead compounds from petrol.
The use of vanadium(V) oxide in the Contact Process
During
the Contact Process for manufacturing sulphuric acid, sulphur dioxide
has to be converted into sulphur trioxide. This is done by passing
sulphur dioxide and oxygen over a solid vanadium(V) oxide catalyst.
  |
| | |
| This
example is slightly different from the previous ones because the gases
actually react with the surface of the catalyst, temporarily changing
it. It is a good example of the ability of transition metals and their
compounds to act as catalysts because of their ability to change their
oxidation state. |
| | |
The
sulphur dioxide is oxidised to sulphur trioxide by the vanadium(V)
oxide. In the process, the vanadium(V) oxide is reduced to vanadium(IV)
oxide.  The vanadium(IV) oxide is then re-oxidised by the oxygen. The vanadium(IV) oxide is then re-oxidised by the oxygen.  This
is a good example of the way that a catalyst can be changed during the
course of a reaction. At the end of the reaction, though, it will be
chemically the same as it started. This
is a good example of the way that a catalyst can be changed during the
course of a reaction. At the end of the reaction, though, it will be
chemically the same as it started. |



- C. G. Frost and L. Mutton, Green Chem., 2010, 12, 1687–1703 .
- M. Irfan, T. N. Glasnov and C. O. Kappe, ChemSusChem, 2011, 4, 300–316
- C. F. Carter, I. R. Baxendale, M. O'Brien, J. P. V. Pavey and S. V. Ley, Org. Biomol. Chem., 2009, 7, 4594–4597 .
- P. McMorn and G. J. Hutchings, Chem. Soc. Rev., 2004, 33, 108–122.
- S.
Ceylan and A. Kirschning, in Recoverable and Recyclable Catalysts, ed.
M. Benaglia, John Wiley & Sons Ltd, 2009, pp. 379–410 .
- A. J. Sandee, D. G. I. Petra, J. N. H. Reek, P. C. J. Kamer and P. W. N. M. Van Leeuwen, Chem.–Eur. J., 2001, 7, 1202–1208
- M. Pagliaro, V. Pandarus, R. Ciriminna, F. Belénd and P. D. Cerà, ChemCatChem, 2012, 4, 432–445 .
- C. W. Kohlpaintner, R. W. Fischer and B. Cornils, Appl. Catal., A, 2001, 221, 219–225
- W. A. Herrmann, C. W. Kohlpaintner, H. Bahrmann and W. Konkol, J. Mol. Catal., 1992, 73, 191
- A. B. Theberge, G. Whyte, M. Frenzel, L. M. Fidalgo, R. C. R. Wootton and W. T. S. Huck, Chem. Commun., 2009, 6225–6227 .
- A. Yoshida, X. Hao and J. Nishikido, Green Chem., 2003, 5, 554–557 .
- E.
Perperi, Y. Huang, P. Angeli, G. Manos, C. R. Mathison, D. J.
Cole-Hamilton, D. J. Adams and E. G. Hope, Dalton Trans., 2004,
2062–2064 .
- S. Liu, T. Fukuyama, M. Sato and I. Ryu, Org. Process Res. Dev., 2004, 8, 477–481
- T. Fukuyama, M. T. Rahman, M. Sato and I. Ryu, Synlett, 2008, 151–163
- J. F. B. Hall, X. Han, M. Poliakoff, R. A. Bourne and M. W. George, Chem. Commun., 2012, 48, 3073–3075 .
- R. A. Bourne, X. Han, M. Poliakoff and M. W. George, Angew. Chem., Int. Ed., 2009, 48, 5322










//////